11.5 Now where does the probability peak?
Clearly the probability density to find an electron at point is
| (11.22) |
but what is the probability to find the electron at a distance from the proton? The key point to realise is that for each value of the electron can be anywhere on the surface of a sphere of radius , so that for larger more points contribute than for smaller . This is exactly the source of the factor in the normalisation integral. The probability to find a certain value of is thus
| (11.23) |
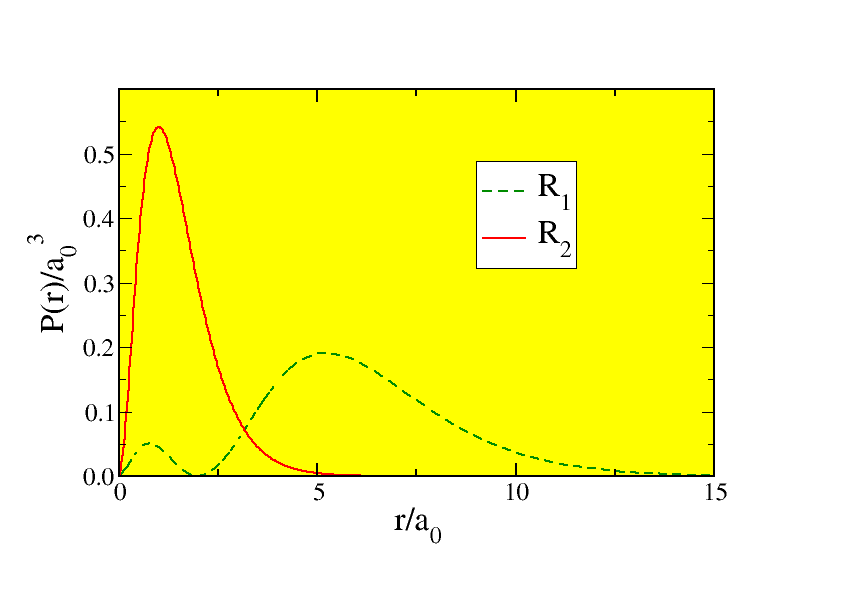
These probabilities are sketched in Fig. 11.2 . The peaks are of some interest, since they show where the electrons are most likely to be found. Let’s investigate this mathematically:
| (11.24) |
if we differentiate with respect to , we get
| (11.25) |
This is zero at . For the first excited state this gets a little more complicated, and we will have to work harder to find the answer.

